Note that the subscript 2 is applied to tell us there are two atoms of oxygen in the diatomic molecule of this element. Shall we do a few examples? If you have oxygen you know that oxygen doesn't hang around by itself. Then you'll have to write up each reactant paying attention to insure you have the right proportions set down for your individual materials. In that case, you'll have to look up that anion or cation and see what elements make it up. This might present a bit of a challenge, paticularly if there is a 'hydroxide' or a 'chlorate' or an 'oxalate' or something like that. Writing a chemical (symbol) equation from a word equation? You'll have to identify the elements and compounds in the equation and snag the correct chemical symbols for each el ement and each compound. 
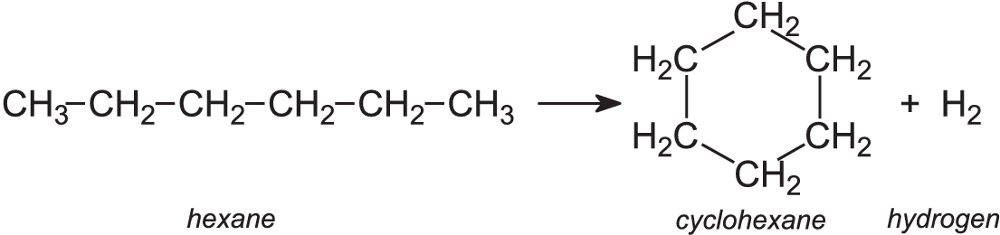
Writing a chemical (symbol) equation from a word equation? You'll have to identify the elements and compounds in the equation and snag the correct chemical symbols. In the realm of global economics and politics, that statement is hard to dispute. Petroleum makes the world go around Displayed formula for thermal cracking of decane.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
